Graphite, as an allotrope of carbon, frequently appears in daily life and industrial production. What is it? Is it a metal, mineral or element? What is it made of? This article will deeply explore the nature of graphite, uncover its layers of secrets. And fully show the unique charm and important value of graphite in the field of science and application.
Table of Contents
ToggleDefinition of graphite
Graphite is a crystalline form of carbon, usually showing a gray-black and opaque solid state, with a unique metallic luster. But sometimes it has another form of it is the amorphous form, which consists ofirreeular arrangements of graphite atoms. Its texture is relatively soft, this softness allows it to leave clear marks on the paper. This property also makes graphite become the main component of pencil lead. In chemical essence, graphite belongs to the allotrope of carbon, which is composed of carbon atoms like diamond, fullerene and other substances. However, the way carbon atoms are arranged in these substances is very different, resulting in huge differences in their physical and chemical properties.
Is Graphite a metal or a mineral?
Graphite is not a metal, but a mineral. Metal usually has good electrical conductivity, thermal conductivity and ductility and other typical characteristics. Although graphite has a certain degree of electrical and thermal conductivity, it does not have the ductility characteristic of metals. Graphite is a naturally occurring product of complex geological processes and meets the definition of a mineral. It exists in specific rocks and deposits in nature. And it is an important form of carbon in the long cycle and evolution process of the earth’s circle, witnessing the movement and change of materials in the earth’s interior.
Is Graphite an element?
Graphite is not an element, but a single substance composed of carbon. An element is a group of atoms that have the same number of nuclear charges (protons). Graphite is a material entity formed by a large number of carbon atoms bound together by specific chemical bonds. This pure substance composed of the same elements is called the elemental. Graphite is a special form of carbon elemental existence. It with a unique crystal structure and physical and chemical properties shows the rich and diverse properties of carbon.
Is Graphite carbon?
Graphite is an allotrope of carbon, composed entirely of the element carbon. Inside the graphite, the carbon atoms are arranged and combined in a very special way to form a unique crystal structure. This structure gives graphite many special properties. So that it and other carbon allotrope such as diamond (known for its hard and transparent properties), fullerene (with a unique spherical or tubular structure) in appearance, physical and chemical properties are significantly different. In many fields they play different roles.
Where Does Graphite Come From?
Natural sources
The natural sources of graphite in nature are more extensive. We can form part of the graphite in metamorphic rocks. For example, in the process of regional metamorphism, the original carbon-containing sediments (such as coal seams) are gradually transformed into graphite through complex metamorphic crystallization under extreme conditions of high temperature and pressure. In addition, some graphite comes from magmatic rocks. And when the magma invades the crust, the carbon carried in the magma crystallizes under specific geological environment and physical and chemical conditions. Thus it forms graphite. Natural graphite deposits are distributed in many countries and regions around the world, among which China, Brazil, India and other countries have relatively rich natural graphite resources. These provide an important material basis for the development of global graphite-related industries.
Artificial graphite
With the rapid development of modern industrial technology, the production of artificial graphite has gradually become an important part of the graphite supply. Artificial graphite is usually produced by high temperature heat treatment of specific carbon-containing raw materials (such as petroleum coke, asphalt coke, etc.). It converts amorphous carbon into graphite. At high temperatures, the non-carbon elements in these carbon-containing raw materials will gradually volatilize. And the carbon atoms will rearrange and crystallize, eventually forming artificial graphite with a structure similar to natural graphite. The production of artificial graphite has a high degree of controllability. And it can precisely control its purity, crystal structure and physical and chemical properties according to different industrial application needs. So that it is widely used in many fields such as steel, batteries, refractories, and so on. This provides a strong support for the development of modern industry.
Graphite types
In summary, graphite can be mainly divided into natural graphite and synthetic graphite two types.
Natural graphite
Natural graphite also includes flake graphite, crystalline graphite and cryptocrystalline graphite.
Flake graphite is characterized by a large and thin flake shape with a wide range of diameter, ranging from a few tenths of a millimeter to several millimeters. These scales have good electrical and thermal conductivity in the plane direction, while their layered structure gives it excellent lubrication properties.
Crystalline graphite crystals are well developed, showing an obvious hexagonal lamellar crystal shape. And its scales are relatively large, and the diameter of the slices is often more than 0.1-0.2 mm. This type of graphite is not abundant in nature. But because of its excellent crystal structure and unique properties, it plays a role in many high-end industrial fields.
The crystal of aphanitic graphite is very small and exists in the form of microcrystalline aggregates. And the specific shape of the crystal can hardly be distinguished by the naked eye. Its fixed carbon content is at a high level, roughly in the range of 60%-80%. In industrial applications, we can use cryptocrystalline graphite as casting and refractory materials.
Synthetic graphite
Synthetic graphite contains three types, each with unique properties and uses.
High purity graphite purity is extremely high, the impurity content is very low, usually more than 99.9%. Its excellent chemical stability and high electrical conductivity make it play a key role in semiconductor manufacturing, high-end chemical industry and other industries with strict purity requirements.
Isostatic pressed graphite is formed by isostatic pressing process and has the characteristics of uniform structure and isotropy. It has excellent mechanical strength, high temperature resistance and thermal shock resistance, excellent performance in metallurgy, EDM and other fields.
Expanded graphite is made of natural graphite by special treatment, with a unique wormlike structure. This can expand rapidly at high temperatures to form a material with excellent thermal insulation and sealing properties. In the field of fireproof materials, expanded graphite can be used to manufacture fireproof seals, fireproof coatings, etc.. It can effectively prevent the spread of fire.
What is Graphite Made of?
Graphite Element
The main constituent element of graphite is carbon. In graphite, carbon atoms are connected to each other by covalent bonds, forming a planar network of hexagons. These planar network structures are stacked layer upon layer in space, forming the unique crystal structure of graphite. Each carbon atom forms a covalent bond with three surrounding carbon atoms. And the presence of this covalent bond gives graphite high stability and strength within the layer. While also laying the foundation for some of its special properties, such as electrical and thermal conductivity.
The chemical formula of graphite
The chemical formula of graphite is usually denoted by C, which clearly indicates that it is composed entirely of carbon. Despite its simple chemical formula, graphite exhibits complex and diverse physical and chemical properties. Due to the unique arrangement of carbon atoms and the existence of many forms of chemical bonding. This characteristic, which is composed of a single element but has rich properties, makes graphite occupy a unique position in the field of materials science. And also making it becomes the focus of numerous research and uses.
Graphite Structure
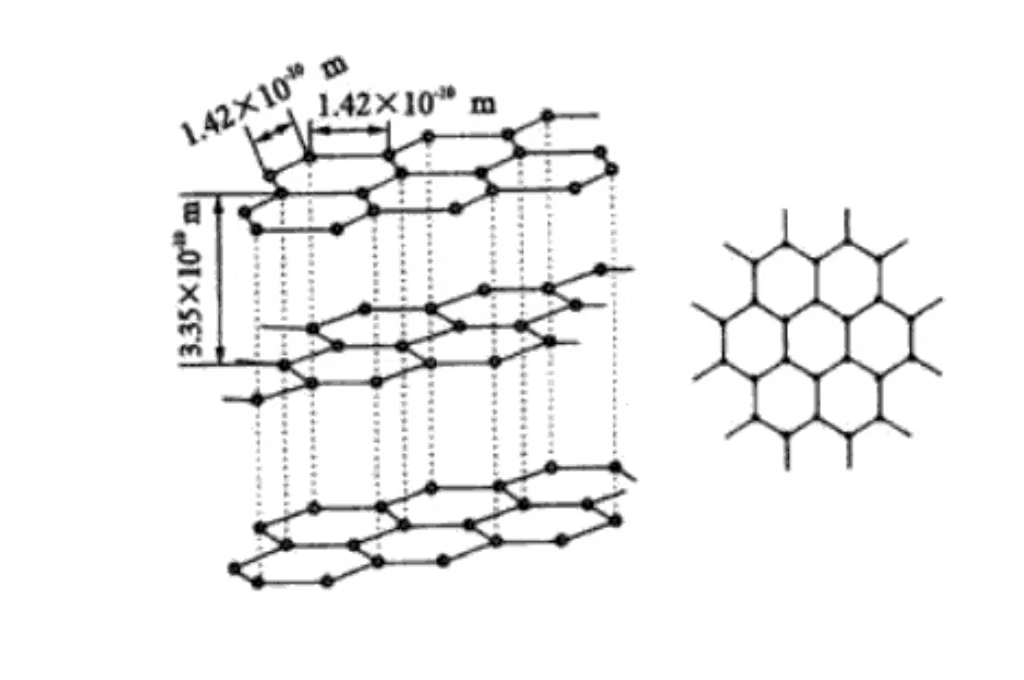
Carbon Arrangement of carbon atom in Graphite
Carbon is arranged in the microstructure of graphite. And the carbon atoms show a remarkable feature of layered arrangement. Each layer of carbon atoms is closely arranged to form a huge hexagonal planar network structure. And the carbon atoms are closely connected through covalent bonds. It makes these plane layers have high stability and strength, and can withstand a certain degree of external force without damage. The layer between the layer is through the weak van der Waals force interaction. This relatively weak interlayer force makes the graphite between the layer and the layer when the small external force is easy to slide, thus giving the graphite good lubrication and flexibility. So that it needs to reduce friction and have a certain deformation ability in the application scenario.
Bonding
The covalent bond between carbon atoms in the graphite layer is a strong chemical bond. It not only ensures the stability and integrity of the graphite layer, but also has a profound impact on the physical properties of graphite. Due to the existence of covalent bonds, electrons are able to move relatively freely between the carbon atoms within the layer. This makes the graphite have good conductive and thermal conductivity within the layer, and can efficiently transfer current and heat.
The van der Waals force between the layers is relatively weak. And its contribution to the physical properties of graphite such as hardness and density is smaller than that of covalent bonds. This synergistic effect of intra-layer covalent bonds and interlayer van der Waals forces creates the unique anisotropic properties of graphite. That is, the physical and chemical properties of graphite in the direction of the layer and the vertical layer are significantly different. And this property needs to be fully considered in material applications to achieve the optimal utilization of graphite properties.
Properties of Graphite
Physical Properties
Color
Graphite usually presents a gray-black appearance color. The formation of this color is closely related to the internal electronic structure of graphite and its absorption and reflection characteristics of light. The carbon atoms in graphite absorb and scatter visible light through specific chemical bonds and electron cloud distribution. So that most of the visible light is absorbed, and only a small amount of light is reflected or scattered. Thus presenting a gray-black visual effect on the macro level. Moreover, the opaque nature of graphite also gives its appearance a unique texture, in sharp contrast to other transparent or translucent materials.
Density
The density of graphite is relatively small, between about 2.09-2.23 g/cm 3. And the specific gravity is also low. This characteristic makes graphite a clear advantage in some application scenarios with strict requirements for weight. For example, in the design of certain components in the aerospace field, if you need to use materials that have a certain degree of conductive property and lubricity, but also can reduce the overall weight. Then graphite becomes a very potential candidate material.
Melting point
Graphite has a very high melting point, about 3652°C -3697 °C. This excellent high-temperature stability allows graphite to maintain its structure and properties relatively stable under extremely high temperature environments. In iron and steel smelting, refractory materials and other high-temperature industrial processes, graphite plays a vital role.
Electrical conductivity
Graphite has excellent electrical conductivity in the layer, which is due to the formation of a stable electron cloud structure between the carbon atoms in the layer through covalent bonds. And electrons can move relatively freely in this structure, so as to achieve efficient conduction of current. At the same time, the thermal conductivity of graphite is also very good, and it can transfer heat quickly.
Lubrication
The lubricating properties of lubricating graphite are due to its unique layered structure. Because the van der Waals force between layers is weak. When the graphite is subjected to external force, the relative sliding between layers is easy to occur. And this sliding process can effectively reduce the coefficient of friction, so as to play a good lubrication role. Whether it is the daily lubrication maintenance of various mechanical equipment in the machinery industry, or the lubrication needs in some special environments (such as high temperature, high pressure or chemical corrosion environment), graphite can show excellent lubrication effects.
Chemical Properties
Graphite corrosion resistance
Graphite has good acid and alkali resistance. It can maintain relatively stable structure and performance in acid and alkali solutions within a certain concentration range. This is because the carbon atoms in graphite form a stable chemical bond energy structure through covalent bonds. This makes the graphite difficult to be destroyed by ions in acid and base solutions. This acid and alkali resistance makes graphite have important application value in some corrosive environments in the chemical industry.
Reactivity with other materials
Under normal temperature conditions, the chemical properties of graphite are relatively stable. And it is not easy to chemically react with most common substances. However, when exposed to high temperature, high pressure or a specific chemical environment, graphite can react with some oxidants (such as oxygen, concentrated sulfuric acid, etc.).
For example, when oxygen is sufficient and the temperature rises to a certain extent, the graphite will undergo oxidation reaction and gradually transform into products such as carbon dioxide. This reactivity limits the application of graphite in some extreme oxidation environments to a certain extent. But it also provides the possibility for some special treatment and modification of graphite.
Thermal expansion coefficient
Graphite has a low coefficient of thermal expansion, which gives it good dimensional stability when the temperature changes. Compared with many other materials, the volume of graphite changes very little during the process of experiencing a larger temperature rise and fall.
In some applications that require high dimensional accuracy of materials, this low coefficient of thermal expansion of graphite is particularly important. It can effectively avoid problems such as component deformation and assembly accuracy reduction caused by temperature fluctuations. So as to ensure the normal operation and stable performance of equipment or instruments in different temperature environments.
Oxidation
Although graphite shows strong oxidation and corrosion resistance at room temperature, under extreme conditions such as high temperature, high humidity or strong oxidation environment, graphite will gradually oxidize and corrosion. For example, long-term exposure to high temperature in the air, the carbon atoms on the surface of graphite will react with oxygen to form an oxide layer.
And over time, the continuous thickening of the oxide layer will lead to changes in the structure and performance of graphite. Such as reduced conductivity and strength. Therefore, in some application areas with high requirements for the oxidation resistance of graphite, it is often necessary to carry out special surface treatment of graphite. Or add antioxidants and other measures. To improve its antioxidant capacity to ensure the performance stability and reliability of graphite materials during use.
Mechanical Properties
Hardness and strength
The strength and hardness of graphite are relatively low, and its Mohs hardness is about 1-2. This property makes graphite relatively easy to shape and process into various shapes during processing. For example, in the manufacture of pencil leads, by mixing graphite and other materials such as clay in different proportions and pressing, it is convenient to make pen leads with different hardness levels to meet different writing needs. However, although the overall strength of graphite is low, it still has a certain strength utilization value in some specific directions. Such as along the direction of the graphite layer.
Elasticity
Due to the unique layered structure of graphite, it shows flexibility and elasticity to a certain extent. When subjected to a small external force, we can bend and deform the layered structure of graphite to a certain extent. And when the external force is removed, we can restore the graphite to the original shape or close to the original shape. This flexibility and elasticity make graphite a potential application prospect in some flexible electronic devices, sealing materials and other emerging fields.
Anisotropy
Anisotropic graphite has extremely obvious anisotropic characteristics. That is, its physical and chemical properties are significantly different in different directions. In terms of conductive property, the conductivity along the graphite layer is much higher than the conductivity in the vertical layer. This is caused by the promotion of covalent bonds on electron conduction and the hindrance of interlayer van der Waals force on electron conduction.
In terms of hardness and strength, the hardness and strength of the vertical layer are relatively high. Because the van der Waals force between the layers limits the relative sliding between the layers to a certain extent. While sliding and deformation are more likely to occur due to the weak interlayer force. This anisotropic characteristic needs special attention and consideration in the application process of graphite. According to the specific application requirements, it is reasonable to select and utilize the properties advantages of graphite in different directions. So as to maximize the utilization of graphite material performance and optimize the application effect.
Thermal and Electrical Properties
There is a close internal relationship between thermal and electrical properties of graphite and thermal properties and excellent performance. Its high thermal conductivity is enough to quickly emit heat, which is of great application value in the field of heat dissipation of electronic equipment. At the same time, graphite’s good conductivity enables it to transmit current efficiently as an excellent conductor in the circuit.
Other Properties
In addition to the many properties mentioned above, graphite has some other special properties. For example, graphite has certain adsorption properties. And its rich pore structure and large specific surface area can absorb some gases and small molecules. This feature has potential application value in gas purification and sewage treatment in the field of environmental protection. By proper modification and treatment of graphite, its adsorption performance can be further improved. It can be used to remove harmful gases in the air (such as formaldehyde, sulfur dioxide, etc.) or heavy metal ions in water, organic pollutants, etc.
6 Uses of Graphite
In Pencils
Graphite is a key component in pencil lead. Due to its soft texture and unique layered structure, it can leave clear marks on paper after mixing and adjusting hardness with clay to meet the needs of writing and painting. From student learning to artistic creation, it is widely used in all kinds of writing tools. This allows people to freely express ideas and creativity.
As a Lubricant
Graphite as a lubricant has good lubricity due to the weak van der Waals force in the middle layer and the interlayer of the layered structure. It is widely used in the mechanical field. Whether it is the internal moving parts of the automobile engine, or the industrial mechanical transmission parts. Or even the aerospace high temperature and high pressure parts, graphite powder can effectively reduce friction, reduce wear. And it also ensures the smooth and efficient operation of the equipment, and extends the service life.
Steel making
Graphite plays an important role in steelmaking. As an electrode, it can introduce current to produce Joule heat melting of scrap steel raw materials. As a carburizer, the carbon content of molten steel can be accurately adjusted. Made of furnace lining material, by virtue of high temperature resistance and corrosion resistance, protect the steelmaking furnace body from high temperature molten steel and slag damage. In addition, graphite has a certain specific heat capacity. It can absorb and release heat during the steelmaking process, helping to regulate temperature fluctuations in the furnace. Effectively support the efficient, safe and stable development of steelmaking process.
Battery
Graphite is of great significance in batteries and is commonly used as a negative electrode material in lithium-ion batteries. Its layered structure provides space for lithium ion embedment and deembedment, charging embedment, discharge discharge, with good conductive property. This ensures the battery charge and discharge cycle. In the research of emerging battery technology, it is also regarded as the basic material. This plays a role in promoting the development of new energy storage.
Refractory materials
Graphite has high melting point and high temperature stability, and is a high-quality refractory material. In metallurgy, ceramics, glass and other industrial high-temperature processes, used in the manufacture of refractory bricks, linings crucibles and so on. It can resist the erosion of metal melt and slag in high temperature environment, maintain structural stability. Reduce heat loss, reduce accident risk, and build a strong safety line for high temperature industrial production.
Nuclear reactors
Graphite serves as a neutron moderator in nuclear reactors. By colliding with neutrons, fast neutrons are slowed down to thermal neutrons. Thus these control the rate of nuclear fission reaction and ensure the stable operation of the reactor. Early nuclear reactors have many uses. But graphite will change under high temperature and neutron irradiation, requiring special technical responses to ensure long-term safe use.
Conclusion
As an allotrope of carbon, graphite has various properties and is widely used. Its various characteristics are related to each other, which determines the performance in different scenarios. From everyday pencils to industrial steelmaking and battery manufacturing to high-tech nuclear reactors, graphite is indispensable. With the development of science and technology, graphite has great potential in emerging fields. And it will occupy a more critical position in materials science, global resource strategy and sustainable development strategy. And it will continue to promote the progress of human society.